A) CH3CH2CH=CHCH2CH=CHCH3
B) CH3CH=CHCH=CHCH2CH3
C) CH2=CHCH2CH2CH2CH=CH2
D) CH2=CHCH=CHCH2CH2CH3
E) CH3CH2CH=C=CHCH2CH3
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Treatment of 4-methylcyclohexene with N-bromosuccinimide in CCl4 would yield mainly: 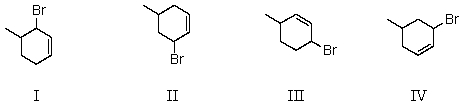
A) I
B) II
C) III
D) IV
E) All of the above
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Essay
The first step in the total synthesis of synthetic cholesterol involves production of the
intermediate structure shown.Which combination of diene and dieneophile will result in
its preparation? 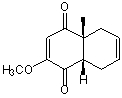
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The LUMO of 1,3-pentadiene has how many electrons in its ground state?
A) 1
B) 2
C) 3
D) 4
E) 0
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How could the following synthesis be carried out? 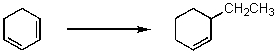
A) (1) Br2/CCl4; (2) CH3CH2MgCl,ether; (3) CH3ONa/CH3OH
B) (1) HBr,80 C; (2) (CH3CH2) 2CuLi,ether
C) (1) HBr,80 C; (2) Mg,ether; (3) CH3CH2OH,then H3O+
D) More than one of the above
E) All of the above
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of these dienes is the most reactive in the Diels-Alder reaction?
A) 1,3-Butadiene
B) 1,4-Pentadiene
C) 2,3,-Dimethyl-1,3-butadiene
D) 1,2-Butadiene
E) 1,4-Cyclohexadiene
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
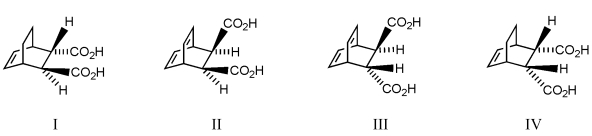
Which diene and dienophile would you choose to synthesize the following compound? 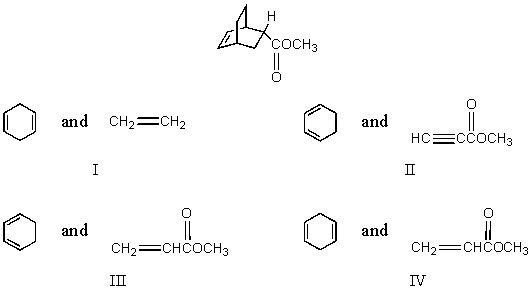
A) I
B) II
C) III
D) IV
E) None of these
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which is the major product of the following reaction? 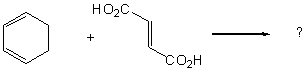
A) I
B) II
C) III
D) IV
E) None of these
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Treatment of 2-butene (cis or trans) with Cl2 at 400 C would yield mainly:
A) CH2ClCHClCH2CH3
B) CH3CHClCH2CH3
C) CH3CH=CClCH3
D) CH3CH=CHCH2Cl and CH3CHClCH=CH2
E) CH3CHClCHClCH3
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which pair does not represent a pair of resonance structures? 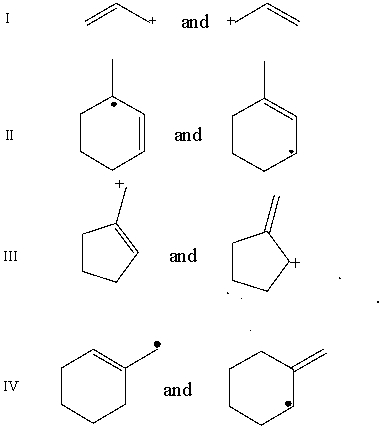
A) I
B) II
C) III
D) IV
E) All of these represent pairs of resonance structures.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Estimate the stabilization energy for 1,3-butadiene using the heats of hydrogenation in Table 1. Table 1. Heats of Hydrogenation for Selected Compounds Compound Moles H2 H(kJ mol-1) 1-Butene 1 -127 1-Pentene 1 -126 1,3-Butadiene 2 -239 Trans-1,3-Pentadiene 2 -226
A) 13 kJ mol-1
B) 15 kJ mol-1
C) 28 kJ mol-1
D) 239 kJ mol-1
E) 112 kJ mol-1
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Ignoring stereochemistry,the 1:1 reaction of bromine with 1,3-cyclohexadiene at 25 C in the dark and in the absence of peroxide forms which of these? 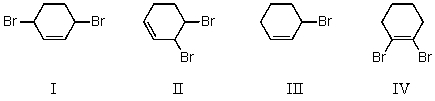
A) I
B) II
C) III
D) IV
E) Both I and II
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which alkene would you expect to have the highest heat of hydrogenation?
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which set of conditions does not result in allylic halogenation of an alkene?
A) Cl2 at 400 C
B) Cl2 in CCl4 at 25 C
C) Cl2,ROOR,h
D) Br2 at low concentration in CCl4
E) N-Bromosuccinimide in CCl4,ROOR
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Short Answer
UV-vis spectroscopy can be used to indicate whether _________________ is present.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following dienes is a cumulated diene?
A) CH2=CHCH2CH2CH=CH2
B) CH2=CHCH=CHCH2CH3
C) CH3CH=C=CHCH2CH3
D) CH3CH=CHCH=CHCH3
E) CH3CH=CHCH2CH=CH2
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A reaction under kinetic (or rate) control will yield predominantly:
A) the most stable product.
B) the product that can be formed in the fewest steps.
C) the product whose formation requires the smallest free energy of activation.
D) the product with the greatest potential energy.
E) the product with the least potential energy.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
An equilibrium-controlled reaction will yield predominantly:
A) the more/most stable product.
B) the product whose formation requires the smallest free energy of activation.
C) the product that can be formed in the fewest steps.
D) the product that is formed at the fastest rate.
E) the product which possesses the greatest potential energy.
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Essay
Steroids are naturally occurring polycyclic compounds classified as lipids.The Diels-
Alder reaction has been widely used in their preparation.Shown below is a precursor
molecule used in a total steroid synthesis.What combination of diene and dieneophile can
be used to prepare this compound through the use of a Diels-Alder cycloaddition reaction. 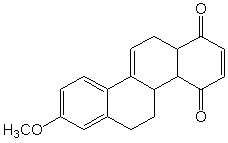
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which carbon-carbon bond in the following compound would you expect to be longest? 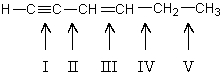
A) I
B) II
C) III
D) IV
E) V
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Showing 141 - 160 of 166
Related Exams