Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of these electron energy level patterns would absorb light with the shortest wavelength? 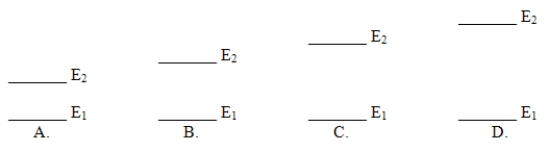
A) A
B) B
C) C
D) D
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many 3d electrons does a V3+ ion have?
A) 6
B) 5
C) 4
D) 3
E) 2
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The correct formula for the dichlorobis(ethylenediamine) chromium(III) ion is
A) [Cr(en) 2Cl2]3+.
B) [Cr(en) Cl2]+.
C) [Cr(en) 2Cl2]2+.
D) [Cr(en) 2Cl2]+.
E) [Cr(en) 3Cl2]+.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Short Answer
What terms describe the geometric isomers that are possible for the complex [CrF2Cl4]3-?
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many 3d electrons does a Mn2+ ion have?
A) 1
B) 2
C) 3
D) 4
E) 5
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the [CoCl6]4- ion.Determine which responses are true statements. I.The oxidation number of Co is -4. II.The coordination number of cobalt is 6. III.It is paramagnetic. IV.It is a low-spin complex.
A) I, II, III, IV
B) I, IV
C) II, III
D) I, III
E) III, IV
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The total number of electrons in the 3d orbitals of a titanium atom is
A) 1
B) 2
C) 3
D) 4
E) 5
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In Na3[Ni(SCN) 5], how many 3d electrons does nickel have?
A) 6
B) 7
C) 8
D) 9
E) 10
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Short Answer
Name the complex ion [Co(H2O)4Cl2]+.
Correct Answer

verified
tetraaquod...View Answer
Show Answer
Correct Answer
verified
View Answer
Short Answer
A molecule or ion that provides an electron pair for coordinate covalent bond formation is called a Lewis ________.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which response gives the correct coordination number (C.N.) and oxidation number (O.N.) of the transition metal atom in [Co(NH3) 2(H2O) 2Cl2]+?
A) C.N.= 2; O.N.= +3
B) C.N.= 3; O.N.= +1
C) C.N.= 4; O.N.= +2
D) C.N.= 6; O.N.= +1
E) C.N.= 6; O.N.= +3
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Showing 81 - 92 of 92
Related Exams